Uribel
23 June, 2023
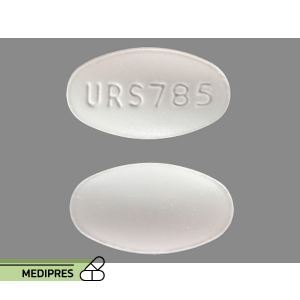
Ursodiol
23 June, 2023Urso
Generic name: Ursodiol
Drug class: Gallstone solubilizing agents
Dosage form: Oral capsule (200 mg; 300 mg; 400 mg), oral tablet (250 mg; 500 mg)
Root of administration: Oral
Dose:
- Gallstone dissolution: 8 to 10 mg/kg orally per day, given in 2 to 3 divided doses
- Gallstone prevention: 300 mg orally 2 times a day (maximum dose: 600 mg/day)
- Primary biliary cirrhosis (PBC): 13 to 15 mg/kg orally per day, given in 2 to 4 divided doses with food
Mechanism of action: Ursodiol reduces cholesterol absorption and is used to dissolve cholesterol gallstones in patients who want an alternative to surgery. It also has multiple mechanisms involved in cholestatic liver diseases, including cytoprotection of the injured bile duct epithelial cells (cholangiocytes) against toxic effects of bile acids, inhibition of apoptosis of hepatocytes, immunomodulatory effects, and stimulation of bile secretion by hepatocytes and cholangiocytes.
Drug usage cases:
- Treatment of gallstones
- Prevention of gallstone formation
- Management of primary biliary cholangitis (PBC)
- Management of primary sclerosing cholangitis
Drug contraindications:
- Complete biliary obstruction
Side effects:
- Abdominal discomfort or pain
- Alopecia (hair loss)
- Diarrhea
- Nausea
- Pruritus (itching)
- Rash
- Leukopenia (low white blood cell count)
- Elevated creatinine or blood glucose levels
- Thrombocytopenia (low platelet count)
Warnings:
- Monitor liver function and bilirubin levels monthly for 3 months after starting therapy, then every 6 months thereafter; consider discontinuing if levels increase significantly with a history of stable liver function
- Monitor and maintain bile flow
- Be cautious in patients with variceal bleeding, hepatic encephalopathy, ascites, and/or need for an urgent liver transplant
- Use with caution in patients with a risk for intestinal stenosis or stasis; withhold and evaluate if obstructive gastrointestinal symptoms occur
- Use during pregnancy only if clearly needed; animal studies have shown no adverse effects on embryo-fetal development at doses greater than human therapeutic doses
- Use during breastfeeding with caution; the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ursodiol and any potential adverse effects on the breastfed child from ursodiol or from the underlying maternal condition
Use during pregnancy or breastfeeding:
Pregnancy: Ursodiol should be used during pregnancy only if clearly needed. Animal studies have shown no adverse effects on embryo-fetal development at doses greater than human therapeutic doses. The estimated background risk of major birth defects and miscarriage in the U.S. general population is 2 to 4% and 15 to 20%, respectively. The potential benefits of treatment should be weighed against the potential risks to the fetus.
Breastfeeding: Ursodiol is naturally present in human milk. There are no reports of adverse effects of ursodiol on the breastfed child, but the reports are extremely limited. There are no data on the effects of ursodiol on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ursodiol and any potential adverse effects on the breastfed child from ursodiol or from the underlying maternal condition.