Jadelle
23 June, 2023
Kalydeco
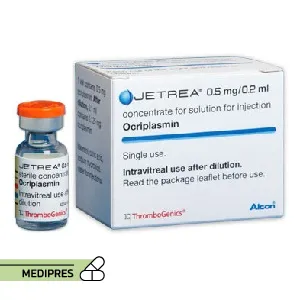
23 June, 2023Jetrea
Generic name: ocriplasmin
Drug class: Miscellaneous ophthalmic agents
Drug class: Ophthalmologic agents (Proteolytic enzymes)
Dosage form: Injection
Route of administration: Intravitreal (injection into the eye)
Dose: Typically, a single 0.125 mg (0.1 mL of 1.25 mg/mL solution) injection into the affected eye.
Mechanism of action: Jetrea contains ocriplasmin, a proteolytic enzyme that cleaves protein components of the vitreous body and vitreoretinal interface, facilitating the resolution of vitreomacular adhesion (VMA) and improving the likelihood of posterior vitreous detachment.
Drug usage cases: Jetrea is used to treat symptomatic vitreomacular adhesion (VMA), a condition where the vitreous gel of the eye adheres to the retina, potentially leading to vision impairment.
Drug contra indications: Jetrea should not be used in patients with known hypersensitivity to ocriplasmin or any component of the formulation. It is also contraindicated in patients with proliferative diabetic retinopathy, retinal detachment, or other conditions that could increase the risk of retinal tears.
Side effects: Common side effects include eye pain, floaters, photophobia, and blurred vision. Serious side effects may include:
- Retinal detachment: Risk of detachment or tear of the retina.
- Reduced visual acuity: Potential for temporary or permanent decrease in vision.
- Intraocular inflammation: Risk of inflammation inside the eye.
- Lens subluxation: Displacement of the eye lens.
Warnings: Jetrea can cause significant changes in vision and retinal structure, necessitating close monitoring by an ophthalmologist. Patients should be informed about the potential risks and signs of retinal detachment. Regular follow-up exams are crucial to assess the treatment outcome and manage any adverse effects.
Use during pregnancy or breastfeeding: The safety of Jetrea during pregnancy is not well established, and it should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is unknown whether ocriplasmin is excreted in human milk, so caution should be exercised when administered to breastfeeding mothers.