Olmesartan
23 June, 2023
Ondansetron
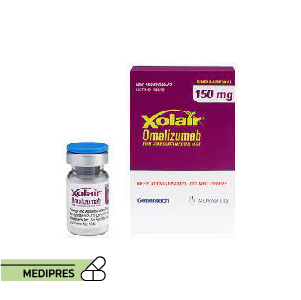
23 June, 2023Omalizumab
Generic name: omalizumab [ OH-ma-LIZ-oo-mab ]
Brand names: Xolair, Xolair Prefilled Syringe
Dosage forms: subcutaneous powder for injection (150 mg), subcutaneous solution (150 mg/mL; 75 mg/0.5 mL)
Drug class: Selective immunosuppressants
Route of administration: Subcutaneous
Available Dose:
- Injectable solution: 150 mg/mL
Mechanism of action: Omalizumab is a monoclonal antibody that binds to immunoglobulin E (IgE), inhibiting its ability to bind to receptors on mast cells and basophils, reducing allergic inflammation.
Drug usage cases: Omalizumab is used for the treatment of moderate to severe persistent asthma in patients with a positive skin test or in vitro reactivity to a perennial aeroallergen, and chronic idiopathic urticaria.
Drug contraindications: Contraindicated in patients with known hypersensitivity to omalizumab or any of its components.
Side effects: Common side effects include injection site reactions, viral infections, upper respiratory tract infections, headache, and sinusitis. Rarely, it may cause severe allergic reactions and anaphylaxis.
Warnings: Caution is advised in patients with a history of anaphylaxis. Patients should be monitored for signs of allergic reactions after administration.
Use during pregnancy or breastfeeding: Omalizumab is categorized as a pregnancy category B drug. It should only be used during pregnancy if needed. It is unknown if omalizumab is excreted in human milk; therefore, caution should be exercised when administered to breastfeeding women.