Sofosbuvir and Velpatasvir
23 June, 2023
Sprintec
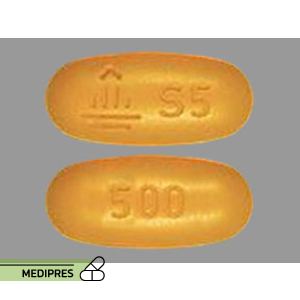
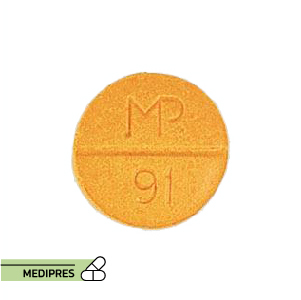
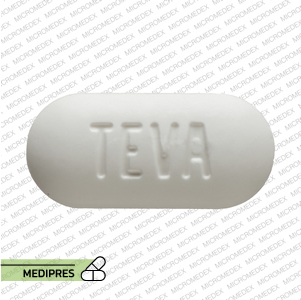
23 June, 2023Spironolactone and hydrochlorothiazide
Generic name: Spironolactone and Hydrochlorothiazide
Drug class: Diuretics (Aldosterone Antagonist and Thiazide)
Dosage form: Oral tablets
Route of administration: Oral
Dose: The recommended adult dose for hypertension is 50 to 100 mg of hydrochlorothiazide and 50 to 100 mg of spironolactone, taken orally once daily or in divided doses. For edema associated with heart failure, cirrhosis, or nephrotic syndrome, the usual adult dose is 25 to 200 mg of hydrochlorothiazide and 25 to 200 mg of spironolactone, taken orally once daily or in divided doses.
Mechanism of action: Spironolactone is an aldosterone antagonist that binds to aldosterone receptors in the distal renal tubules, inhibiting sodium and water reabsorption while promoting potassium retention. Hydrochlorothiazide is a thiazide diuretic that inhibits the sodium-chloride cotransporter in the distal convoluted tubules, leading to increased excretion of sodium, chloride, and water, thereby reducing blood volume and blood pressure.
Drug usage cases:
- Hypertension (high blood pressure)
- Edema (fluid retention) associated with heart failure, cirrhosis of the liver, or nephrotic syndrome
- Hypokalemia (low potassium levels) induced by other diuretics
Drug contraindications:
- Anuria (inability to urinate)
- Acute renal insufficiency
- Significant impairment of renal excretory function
- Hypercalcemia (high blood calcium levels)
- Hyperkalemia (high blood potassium levels)
- Addison’s disease (adrenal gland disorder)
- Allergy to sulfonamide-derived drugs (e.g., thiazide diuretics)
- Hypersensitivity to spironolactone or hydrochlorothiazide
Side effects:
- Electrolyte imbalances, including hyperkalemia (high potassium levels) and hyponatremia (low sodium levels)
- Hypotension (low blood pressure) leading to dizziness, lightheadedness, or fainting
- Dehydration symptoms such as increased thirst, dry mouth, and dark yellow or brown urine
- Gout attacks characterized by severe pain, redness, warmth, or swelling in joints, particularly the big toe
- Kidney dysfunction signs like decreased urine output, swelling in ankles, hands, or feet
- Menstrual irregularities in women, including amenorrhea (absence of menstruation) and metrorrhagia (intermenstrual bleeding)
- Gynecomastia (breast enlargement) in men
- Photosensitivity (increased sensitivity to sunlight)
- Skin rashes or itching
- Elevated blood sugar levels (hyperglycemia)
Warnings:
- Monitor electrolyte levels regularly, especially potassium and sodium
- Use caution in patients with a history of kidney or liver disease
- Avoid concurrent use with other potassium-sparing diuretics, ACE inhibitors, or angiotensin II receptor antagonists due to increased risk of hyperkalemia
- Discontinue use if signs of severe allergic reactions occur, such as difficulty breathing, swelling of the face or throat, or severe skin reactions
- Use with caution in patients with a history of gout
- Assess blood pressure regularly to prevent hypotension
- Monitor for signs of dehydration, including excessive thirst, dry mouth, and dark urine
- Be aware of potential drug interactions with medications like lithium, NSAIDs, and other diuretics
Use during pregnancy or breastfeeding: The safety of spironolactone and hydrochlorothiazide during pregnancy has not been established. Animal studies have shown potential risks, but human data is limited. Therefore, this medication should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is not known whether spironolactone and hydrochlorothiazide are excreted in human breast milk. Due to the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.